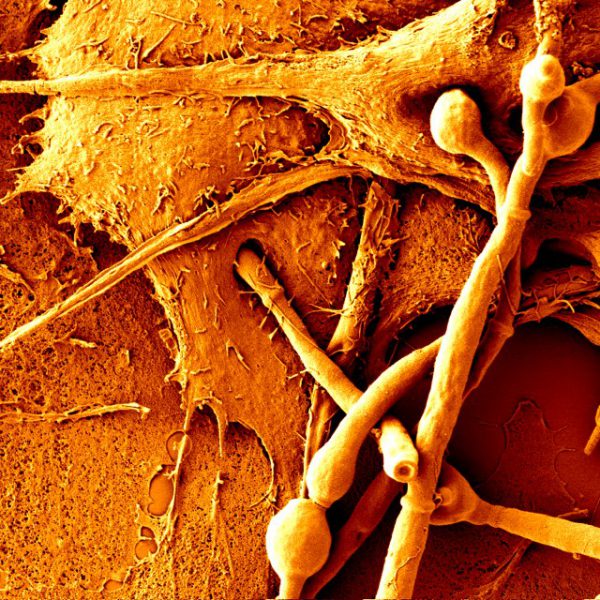
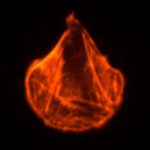
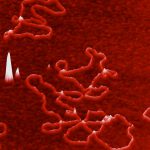
S. cerevisiae cells can form multicellular clumps under adverse growth conditions that protect cells from harsh environmental stresses. The floc formation is based on the self-interaction of Flo proteins via an N-terminal PA14 lectin domain. We have focused on the flocculation mechanism and its role. We found that carbohydrate specificity and affinity are determined by the accessibility of the binding site of the Flo proteins where the external loops in the ligand-binding domains are involved in glycan recognition specificity. We demonstrated that, in addition to the Flo lectin-glycan interaction, glycan-glycan interactions also contribute significantly to cell-cell recognition and interaction. Flocculation provides a uniquely organised multicellular ultrastructure that is suitable to induce and accomplish cell mating. Therefore, flocculation is an important mechanism to enhance long-term yeast survival.
Microbial pathogens may express a wide range of carbohydrate-specific adhesion proteins that mediate adherence to host tissues. Pathogen attachment to host cells is achieved through the binding of these lectin-like adhesins to glycans on human glycoproteins. In only a few cases have the human receptors of pathogenic adhesins been described. We developed a new strategy to predict these interacting receptors. Therefore, we developed a novel LGI network that would allow the mapping of potential adhesin binding receptors in the host with prioritisation, based on the experimental binding data, of the most relevant interactions. New potential targets for the selected adhesins (bacterial uroepithelial FimH from E. coli and fungal Epa and Als adhesins from C. glabrata and C. albicans) were predicted and experimentally confirmed. This methodology was also used to predict lectin interactions with human-pathogenic viruses and to discover whether FimH adhesin has anti-HIV activity.
Cystic fibrosis (CF) is currently the most common, lethal genetic disorder among the Caucasian population. Patients with CF are very susceptible to bacterial chronic airway infections such as S. maltophilia. Treatment with the antibiotic ciprofloxacin leads to the production of three distinct populations of MVs: the classic outer membrane vesicles (OMVs), larger vesicles containing both inner and outer membrane (so called inner-outer membrane vesicles (IOMVs)), and IOMVs that are enriched with fimbriae. This project aims to further structurally characterise the OMVs.